高等反应工程习题7有待改进.docx
《高等反应工程习题7有待改进.docx》由会员分享,可在线阅读,更多相关《高等反应工程习题7有待改进.docx(15页珍藏版)》请在冰豆网上搜索。
高等反应工程习题7有待改进
高等反应工程习题7(例4-1)
本小组(第7组)的催化剂活性校正系数COR取0.380。
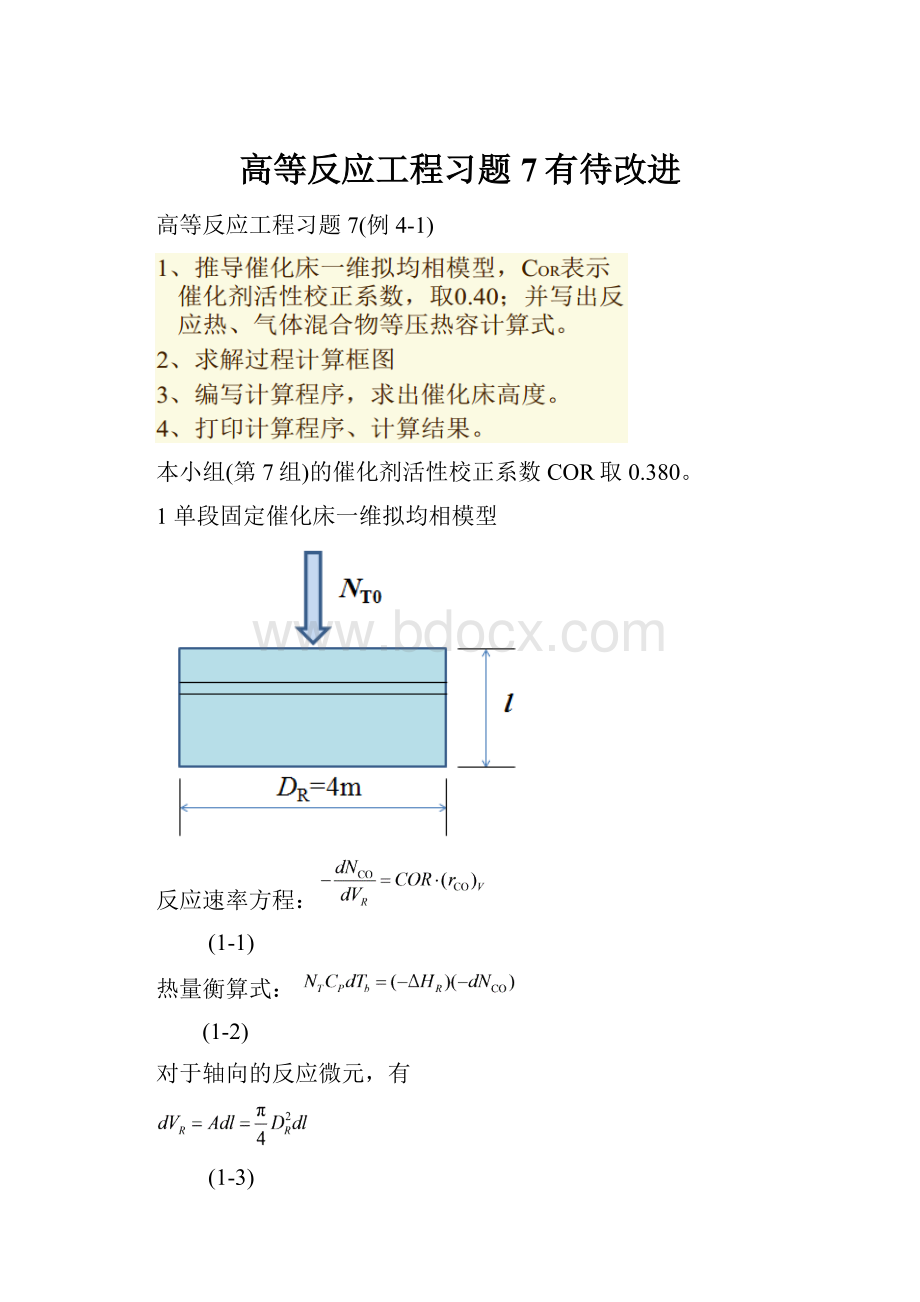
1单段固定催化床一维拟均相模型
反应速率方程:
(1-1)
热量衡算式:
(1-2)
对于轴向的反应微元,有
(1-3)
(1-4)
对于一氧化碳变换反应,有
(1-5)
此外,
(1-6)
由式(1-1)~式(1-6),经过整理可以得到
(1-7)
(1-8)
几个重要公式:
本征速率方程
(1-9)
式(1-9)中,
(1-10)
(1-10)
反应热
(1-11)
混合气体等压摩尔热容
(1-12)
(1-13)
(1-14)
(1-15)
(1-16)
(1-17)
2求解过程计算框图
求解问题:
给定进口温度(371℃)和出口一氧化碳含量(0.0212),求催化床高度。
2.1逐步扫描法求催化床高度
2.2二分法求催化床高度
3编写计算程序,求出催化床高度
运行环境:
MatlabR2011a
3.1六个函数文件:
函数文件
(1)~(5)在两个主程序中都有用到,函数文件(6)在二分法的主程序中用到。
(1)rw.m
%-Rgistheuniversalgasconstant£¬8.314J/K/mol
function[rw1]=rw(Tb,yCO)
pCO=yCO.*3.05;pCO2=(0.1298-yCO).*3.05;pH2=(0.4345-yCO).*3.05;pH2O=(yCO+0.2925).*3.05;
rw1=kT(Tb).*pCO.*pCO2.^-0.5.*(1-pCO2.*pH2./(Kp(Tb).*pCO.*pH2O));%Calculatereactionrate,unit:
kmol/kg/h
functionkT1=kT(Tb)
Rg=8.314;
kT1=3.08.*10.^6.*0.101325.^-0.5.*exp(-104600./(Rg.*Tb));%CalculatekT
end
functionKp1=Kp(Tb)
Kp1=exp(2.3026.*(2185./Tb-0.1102./2.3026.*log(Tb)+0.6218.*10.^-3.*Tb-1.0604.*10.^-7.*Tb.^2-2.218));%CalculateequilibriumconstantKp
end
end
(2)HrCO.m
function[Hr]=HrCO(Tb)
Hr=-(10000+0.219.*Tb-2.845.*10.^-3.*Tb.^2+0.9703.*10.^-6.*Tb.^3).*4.184;%Calculatetheheatofreaction
end
(3)cpM.m
function[cp]=cpM(yCO,Tb)
cp=yCO.*cpCO(Tb)+(yCO+0.2925).*cpH2O(Tb)+(0.1298-yCO).*cpCO2(Tb)+(0.4345-yCO).*cpH2(Tb)+0.1432.*cpN2(Tb);%Calculatemixedgasisobaricmolarheatcapacity
functioncp1CO=cpCO(Tb)
pCO=yCO.*3.05;
cp1CO=(3.86771-0.23279.*(Tb/100)+0.046135.*(Tb/100).^2-0.2168.*10.^-2.*(Tb/100).^3+0.42112.*10.^-2.*(pCO./0.101325)-0.4694.*10^-3.*(pCO./0.101325).*(Tb./100)).*8.314;
end
functioncp2H2O=cpH2O(Tb)
pH2O=(yCO+0.2925).*3.05;
cp2H2O=(0.65765-0.049712.*(Tb/100)+0.5269.*10.^-3.*(Tb./100).^-3+0.020739.*(pH2O./0.101325)-0.27123.*10^-2.*(pH2O./0.101325).*(Tb./100)).*18.0513.*4.184;
end
functioncp3CO2=cpCO2(Tb)
pCO2=(0.1298-yCO).*3.05;
cp3CO2=(3.18266+0.53754.*(Tb./100)-0.020125.*(Tb./100).^2+0.018520.*(pCO2./0.101325)-0.22009.*10.^-2.*(pCO2./0.101325).*(Tb/100)).*8.314;
end
functioncp4H2=cpH2(Tb)
pH2=(0.4345-yCO).*3.05;
cp4H2=(6.8712+0.03135.*(Tb/100)+0.14138.*10.^-2.*(pH2./0.101325)-0.6.*10.^-6.*(pH2./0.101325).^2+0.1603.*10.^-3.*(pH2./0.101325).*(Tb/100)).*4.184;
end
functioncp5N2=cpN2(Tb)
pN2=0.1432.*3.05;
cp5N2=(4.23329-0.4145.*(Tb./100)+0.072309.*(Tb./100).^2-0.34116.*10.^-2.*(Tb./100).^3+0.57726.*10.^-2.*(pN2./0.101325)-0.7404.*10.^-3.*(pN2./0.101325).*(Tb./100)).*8.314;
end
end
4)dyCOdl.m
%-DRisthefixedbeddiameter,unit:
m
%-pbisthecatalystbulkdensity,unit:
kg/m3
%-CORisthecatalystcorrectioncoefficient£¬hereCORis0.380.
%-NT0isnletgasflow,unit:
kmol/h
%-piiscircumferenceratio
function[dyCOdl1]=dyCOdl(yCO,Tb)
DR=4;pb=1500;COR=0.380;NT0=9707.4;
dyCOdl1=pi./4.*DR.^2.*pb.*COR./NT0.*(-rw(Tb,yCO));%CalculatedyCO/dl
end
5)dTbdl.m
function[dTbdl1]=dTbdl(yCO,Tb)
dTbdl1=HrCO(Tb)./cpM(yCO,Tb).*dyCOdl(yCO,Tb);%CalculatedTb/dl
end
6)RKS4yCO.m
function[e]=RKS4yCO(L)
yCO=[];Tb=[];
yCO
(1)=0.0810;Tb
(1)=644.15;
h=L./20;
fori=1:
20
k1=feval('dyCOdl',yCO(i),Tb(i));t1=feval('dTbdl',yCO(i),Tb(i));%UsingRunge-Kutta'smethodtocalculateyCOandTb
k2=feval('dyCOdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));t2=feval('dTbdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));
k3=feval('dyCOdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));t3=feval('dTbdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));
k4=feval('dyCOdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));t4=feval('dTbdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));
yCO(i+1)=yCO(i)+h.*(k1+2.*k2+2.*k3+k4)./6;%CalculateexityCOofthegivenlenth
Tb(i+1)=Tb(i)+h.*(t1+2.*t2+2.*t3+t4)./6;
end
e=yCO(i+1)-0.0212;
end
3.2主程序与运行结果
3.2.1逐步扫描法
主程序:
calculateyCOTb.m
%-yCOisthecarbonmonoxidemolarfraction
%-Tbisthebedtemperature,unit:
K
%-DeltHrCOistheheatofreaction,unit:
J/mol
%-cpMixisthemixedgasisobaricmolarheatcapacity,unit:
J/K/mol
%-Listheheightofthebed,unit:
m
yCO=[];Tb=[];L=[];DeltHrCO=[];cpMix=[];
yCO
(1)=0.0810;Tb
(1)=644.15;L
(1)=0;h=0.02;
DeltHrCO
(1)=HrCO(Tb
(1));cpMix
(1)=cpM(yCO
(1),Tb
(1));
i=1;
whileyCO(i)-0.0212>0.00001
k1=feval('dyCOdl',yCO(i),Tb(i));t1=feval('dTbdl',yCO(i),Tb(i));%UsingRunge-Kutta'smethodtocalculateyCOandTb
k2=feval('dyCOdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));t2=feval('dTbdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));
k3=feval('dyCOdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));t3=feval('dTbdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));
k4=feval('dyCOdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));t4=feval('dTbdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));
yCO(i+1)=yCO(i)+h.*(k1+2.*k2+2.*k3+k4)./6;Tb(i+1)=Tb(i)+h.*(t1+2.*t2+2.*t3+t4)./6;
L(i+1)=L(i)+h;
DeltHrCO(i+1)=HrCO(Tb(i+1));
cpMix(i+1)=cpM(yCO(i),Tb(i));
i=i+1;
end
R=[L',(Tb'-273.15),Tb',yCO',DeltHrCO',cpMix'];
disp('------------------------------------------------------------------------------------------')
formatshortg
disp('床高´床层温度床层温度一氧化碳分率反应热混合气体等压摩尔热容')
disp('l/mtb/℃tb/KyCODeltHr/(J/mol)CpMix/(J/mol/K)')
disp('------------------------------------------------------------------------------------------')
disp(R)
disp('------------------------------------------------------------------------------------------')
主程序运行结果:
…
可以发现,床层温度为4.32m时,出口一氧化碳摩尔分率达到0.0212,满足给定要求。
3.2.2二分法
通过试算,发现当床层高度为2m时,
,当床层高度为5m时,
,因此,选取床层高度的计算范围为[2,5]。
主程序:
dichotomycalculateL.m
a=2;b=5;
whileabs(b-a)>0.0001
x=(b+a)./2;
ifRKS4yCO(x)==0
break
elseifsign(RKS4yCO(x))==sign(RKS4yCO(b))
b=x;
else
a=x;
end
end
yCO=[];Tb=[];
yCO
(1)=0.0810;Tb
(1)=644.15;
h=x./20;DR=4;
fori=1:
20
k1=feval('dyCOdl',yCO(i),Tb(i));t1=feval('dTbdl',yCO(i),Tb(i));%UsingRunge-Kutta'smethodtocalculateyCOandTb
k2=feval('dyCOdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));t2=feval('dTbdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));
k3=feval('dyCOdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));t3=feval('dTbdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));
k4=feval('dyCOdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));t4=feval('dTbdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));
yCO(i+1)=yCO(i)+h.*(k1+2.*k2+2.*k3+k4)./6;
Tb(i+1)=Tb(i)+h.*(t1+2.*t2+2.*t3+t4)./6;
DeltHrCO(i+1)=HrCO(Tb(i+1));
cpMix(i+1)=cpM(yCO(i),Tb(i));
end
R=[371,Tb(i+1),0.0810,yCO(i+1),x,pi./4.*DR.^2.*x];
disp('-------------------------------------------------------------------------------------------')
disp('进口温度出口温度进口一氧化碳出口一氧化碳床层高度催化剂体积(COR为0.380)')
disp('/℃/℃摩尔分率摩尔分率/m/m3')
disp('-------------------------------------------------------------------------------------------')
disp(R)
disp('-------------------------------------------------------------------------------------------')
主程序运行结果:
可以发现,当床层高度为4.3206m时,出口一氧化碳摩尔分率为0.0212,满足给定要求。
二分法与逐步扫描法的运算结果相近。
4给定出口一氧化碳含量(0.0212),确定最佳进口温度(选做)
试算时,发现进口温度超过一定数值时,龙格库塔法求解yCO和Tb过程中的k1和t1的值极小,导致计算陷入死循环。
改变进口温度并经过多次试算后,确定进口温度计算范围的终点取660.15K,进口温度的始点这里取638.15K。
由于催化剂体积对进口温度的一阶微分方程不容易获得,本题的求解思路是通过改变进口温度得到相应的催化剂体积,通过对运算结果列表的观察确定最佳进口温度。
3.1的中函数文件
(1)~(5)在下面的主程序中有用到。
主程序:
calculatebestTb.m
yCO=[];Tb=[];L=[];
V=[];Lenth=[];Tin=[];Tout=[];yCOin=[];yCOout=[];
yCO
(1)=0.0810;L
(1)=0;h=0.02;
j=1;
forT=638.15:
1:
660.15
Tb
(1)=T;i=1;DR=4;
whileyCO(i)-0.0212>0.00001
k1=feval('dyCOdl',yCO(i),Tb(i));t1=feval('dTbdl',yCO(i),Tb(i));%UsingRunge-Kutta'smethodtocalculateyCOandTb
k2=feval('dyCOdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));t2=feval('dTbdl',(yCO(i)+h./2.*k1),(Tb(i)+h./2.*t1));
k3=feval('dyCOdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));t3=feval('dTbdl',(yCO(i)+h./2.*k2),(Tb(i)+h./2.*t2));
k4=feval('dyCOdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));t4=feval('dTbdl',(yCO(i)+h.*k3),(Tb(i)+h.*t3));
yCO(i+1)=yCO(i)+h.*(k1+2.*k2+2.*k3+k4)./6;Tb(i+1)=Tb(i)+h.*(t1+2.*t2+2.*t3+t4)./6;
L(i+1)=L(i)+h;
DeltHrCO(i+1)=HrCO(Tb(i+1));
cpMix(i+1)=cpM(yCO(i),Tb(i));
i=i+1;
end
Tin(j)=T;Tout(j)=Tb(i);
yCOin(j)=0.0810;yCOout(j)=yCO(i);
Lenth(j)=L(i);V(j)=pi./4.*DR.^2.*L(i);
j=j+1;
end
formatshortg
R=[(Tin'-273.15),(Tout'-273.15),yCOin',yCOout',Lenth',V'];
disp('------------------------------------------------------------------------------------------')
disp('进口温度出口温度进口一氧化碳出口一氧化碳床层高度催化剂体积(COR为0.380)')
disp('/℃/℃摩尔分率摩尔分率/m/m3')
disp('------------------------------------------------------------------------------------------')
disp(R)
disp('------------------------------------------------------------------------------------------')
主程序的运算结果:
可以发现,在进口温度为365~387℃的范围内,催化剂体积随进口温度的升高而下降。
当催化剂体积当进口温度为383℃时,催化剂的体积最小,为46.998m3。